As a severe supply crunch forces hotels and hostels across cities like Hyderabad and Bengaluru to suspend operations, the critical role of Liquefied Petroleum Gas (LPG) is taking center stage. This versatile fuel, primarily a mix of propane and butane, is naturally gaseous at room temperature. It earns the “liquefied” title because it is pressurized into a compact liquid for efficient transport in those familiar steel cylinders found in our kitchens. When the pressure is released, the liquid rapidly turns back into gas and burns efficiently.
An Accidental Discovery
The discovery of LPG was a serendipitous byproduct of a problem. In 1910, American chemist Walter Snelling investigated why gasoline in a car’s tank evaporated so quickly. He discovered that “unstable” gases—propane and butane—were the culprits escaping into the air. By 1912, Snelling developed a method to capture and condense these gases, turning a “waste” product of the oil industry into a portable energy revolution.
Production and Safety
LPG primarily comes from natural gas processing (especially ‘wet’ gas) and crude oil refining, with splits varying by region (often ~50-60% from gas).
In its pure form, LPG is entirely colorless and odorless. This makes it difficult to detect. To solve this, suppliers add a potent chemical called Ethyl Mercaptan (also known as Ethanethiol). This additive provides the distinctive “rotten egg” or “sulfur” scent, ensuring users can immediately detect even a minor leak, and prevent explosions.
The Science of the Cylinder
You will never see LPG stored in square or rectangular containers because of the physics of pressure. When a gas is liquefied under pressure, it exerts equal force in all directions. Square containers have weak points at the corners and joints where stress concentrates, making them prone to structural failure or explosions. Cylindrical shapes distribute this internal pressure uniformly across the curved surface, providing maximum structural integrity and safety.
India’s Paradox: Major Producer, Major Importer
Globally, the production landscape is dominated by energy-rich nations. As of 2026, the United States remains the world’s largest producer, followed by China, Russia, India, and Saudi Arabia. India is therefore among the major LPG-producing countries, thanks to its extensive refining network.
Yet domestic production meets only about 40 percent of the country’s demand. With hundreds of millions of households using LPG for cooking, consumption has surged far beyond what local refineries and gas fields can supply. The shortfall is bridged through imports, which now account for roughly 60 percent of India’s LPG requirement.
Most of these shipments originate in the Gulf region, particularly from producers such as Saudi Arabia, Qatar, and United Arab Emirates.
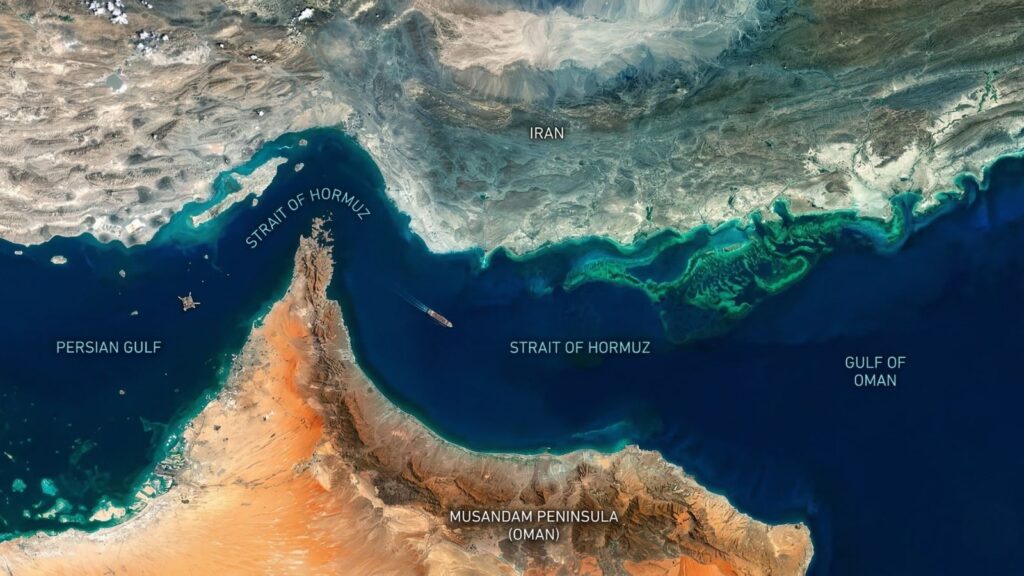
Why the Strait of Hormuz Plays a Key Role
Ships carrying LPG from the Gulf to India must pass through the narrow Strait of Hormuz, one of the world’s most critical energy chokepoints. Nearly a fifth of global oil and a significant share of LPG shipments transit this corridor.
Because the strait lies between Iran and Oman, vessels passing through are closely monitored by Iranian naval forces. During periods of geopolitical tension in West Asia, tankers moving through the strait often sail under heightened scrutiny, with naval patrols keeping a watchful eye on energy cargoes moving toward major importers such as India.
This fragile geography means that any disruption along this narrow maritime corridor can quickly ripple across Asian energy markets, explaining why even distant conflicts can lead to LPG shortages thousands of kilometers away.



